The Polypeptide Chain Passes Through An Aqueous Pore In The Translocator
It has long been debated whether chains are transferred across the in direct contact with the or through a pore in a . The debate ended with the purification of the protein translocator, which was shown to form a water-filled pore in the membrane through which the polypeptide chain traverses the membrane. The translocator, called the Sec61 , consists of three or four protein complexes, each composed of three transmembrane proteins, that assemble into a donutlike structure.
When a binds, the central hole in the translocator lines up with a tunnel in the large ribosomal through which the growing chain exits from the ribosome . The bound ribosome forms a tight seal with the translocator, such that the space inside the ribosome is continuous with the of the and no molecules can escape from the ER . The pore in the translocator cannot be open permanently, however if it were, Ca2+ would leak out of the ER when the ribosome detaches. It is thought that a lumenal ER serves as a plug or that the translocator itself can rearrange to close the pore when no ribosome is bound. Thus, the pore is a dynamic structure that opens only transiently when a ribosome with a growing polypeptide chain attaches to the ER .
Evidence for a continuous aqueous pore joining the ER lumen and the interior of the ribosome. In this experiment, a fluorescent dye is attached to a portion of the growing polypeptide chain that is still contained within the ribosome. In free ribosomes,
Some Membrane Proteins Acquire A Covalently Attached Glycosylphosphatidylinositol Anchor
As discussed in Chapter 10, several cytosolic enzymes catalyze the covalent addition of a single chain or prenyl group to selected proteins. The attached lipids help to direct these proteins to cell membranes. A related process is catalyzed by enzymes, which covalently attach a glycosylphosphatidyl-inositol anchor to the of some proteins destined for the . This forms in the of the ER, where, at the same time, the transmembrane segment of the is cleaved off . A large number of plasma membrane proteins are modified in this way. Since they are attached to the exterior of the plasma membrane only by their GPI anchors, they can in principle be released from cells in soluble form in response to signals that activate a specific phospholipase in the plasma membrane. Trypanosome parasites, for example, use this mechanism to shed their coat of GPI-anchored surface proteins if attacked by the . GPI anchors are also used to direct plasma membrane proteins into rafts and thus segregate the proteins from other membrane proteins, as we discuss in Chapter 13.
The attachment of a GPI anchor to a protein in the ER. Immediately after the completion of protein synthesis, the precursor protein remains anchored in the ER membrane by a hydrophobic C-terminal sequence of 1520 amino acids the rest of the
Where Do The Amino Acids Come From
The amino acids in our body come from the food we eat. We also make them in our body. For example, other animals make proteins and we eat those. Our bodies take that chain and break it down into the individual amino acids. Then it can remake them into any protein that we need.
Once the proteins are broken down into amino acids in the digestive system, they are taken to our cells and kind of float around inside the cell, as those little individual beads in our analogy. And then inside the cell, your body basically connects them together to make the proteins that your body needs to make.
We can make about half of the amino acids we need on our own, but we have to get the others from our food.
Read Also: What Is The Most Protein Dense Food
Specific Enzymes Couple Each Amino Acid To Its Appropriate Trna Molecule
We have seen that, to read the in , cells make a series of different tRNAs. We now consider how each becomes linked to the one in 20 that is its appropriate partner. Recognition and attachment of the correct amino acid depends on enzymes called , which covalently couple each amino acid to its appropriate set of tRNA molecules . For most cells there is a different synthetase for each amino acid one attaches glycine to all tRNAs that recognize codons for glycine, another attaches alanine to all tRNAs that recognize codons for alanine, and so on. Many bacteria, however, have fewer than 20 synthetases, and the same synthetase enzyme is responsible for coupling more than one amino acid to the appropriate tRNAs. In these cases, a single synthetase places the identical amino acid on two different types of tRNAs, only one of which has an that matches the amino acid. A second enzyme then chemically modifies each incorrectly attached amino acid so that it now corresponds to the anticodon displayed by its covalently linked tRNA.
The structure of the aminoacyl-tRNA linkage. The carboxyl end of the amino acid forms an ester bond to ribose. Because the hydrolysis of this ester bond is associated with a large favorable change in free energy, an amino acid held in this way is said
Protein And Weight Control
Eating protein-rich foods has been shown to increase our feeling of fullness more than foods high in fat or carbohydrate. There is good evidence from short term studies that diets high in protein can help reduce overall calorie intake and prompt weight loss. 5 However, the evidence for long-term weight maintenance is less clear.5 Like all diets, a high protein diet is only effective if it is stuck to, which can be difficult for some people and low adherence may partly explain the limited benefit observed for long term weight maintenance.5
You May Like: Premier Protein Shake – Cake Batter Delight
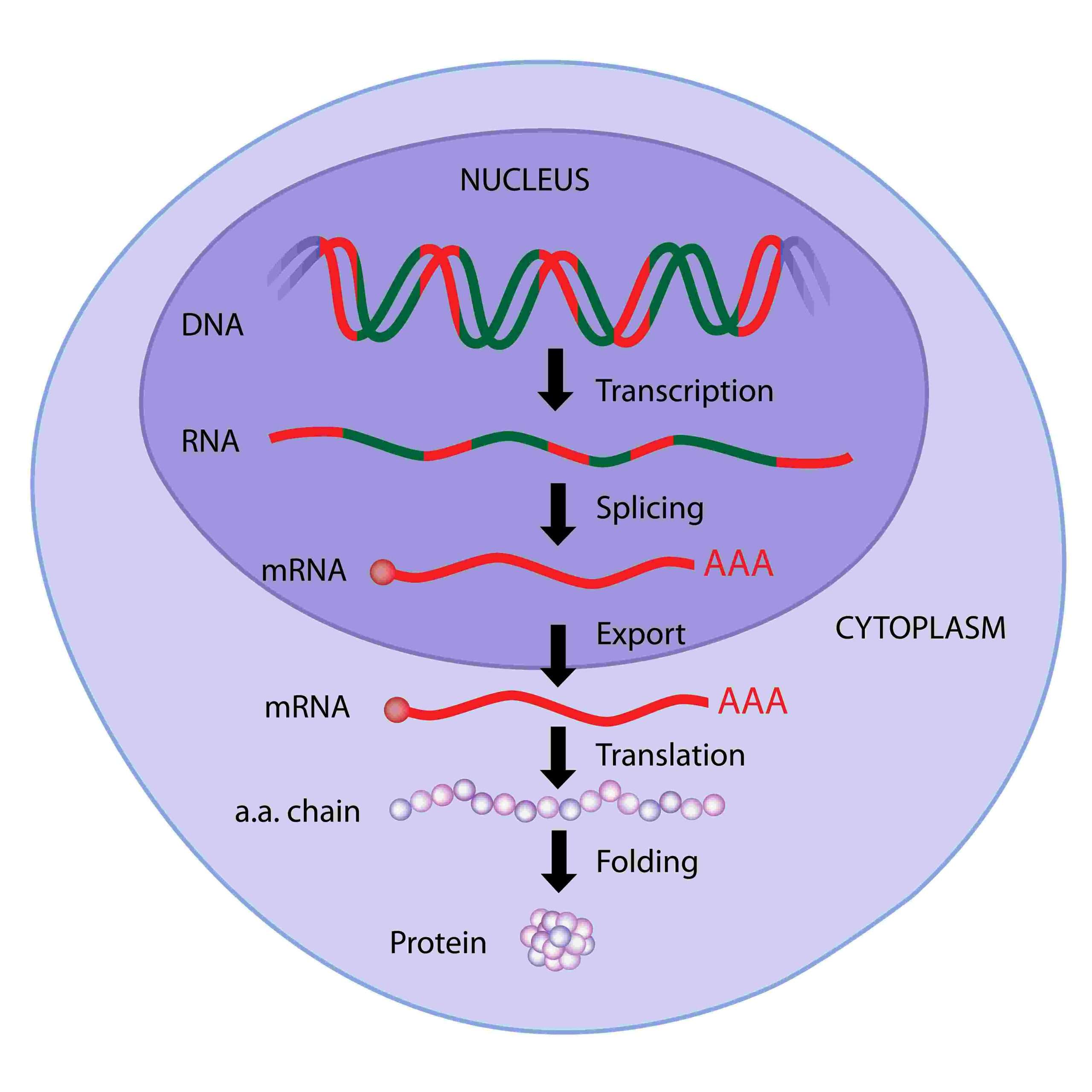
There Are Many Steps From Dna To Protein
We have seen so far in this chapter that many different types of chemical reactions are required to produce a properly folded from the information contained in a . The final level of a properly folded protein in a cell therefore depends upon the efficiency with which each of the many steps is performed.
What Is The Structure Of Proteins
Proteins contain up to four different orders of structure. The string of amino acids is the proteins primary structure. Interactions between the different amino acids cause certain regions of the polypeptide chain to fold into stable patterns called a secondary structure. Examples of secondary structure include alpha helices or beta sheets. These secondary structures can in turn interact with each other, giving rise to the tertiary structure.
And finally, there are times when more than one copy of a protein has to work together to get its job done. In these cases, each of the proteins is referred to as a subunit. The quaternary structure of a protein is the final structure of all the subunits together.
Related articles
Recommended Reading: Gatorade Zero Protein Nutrition Facts
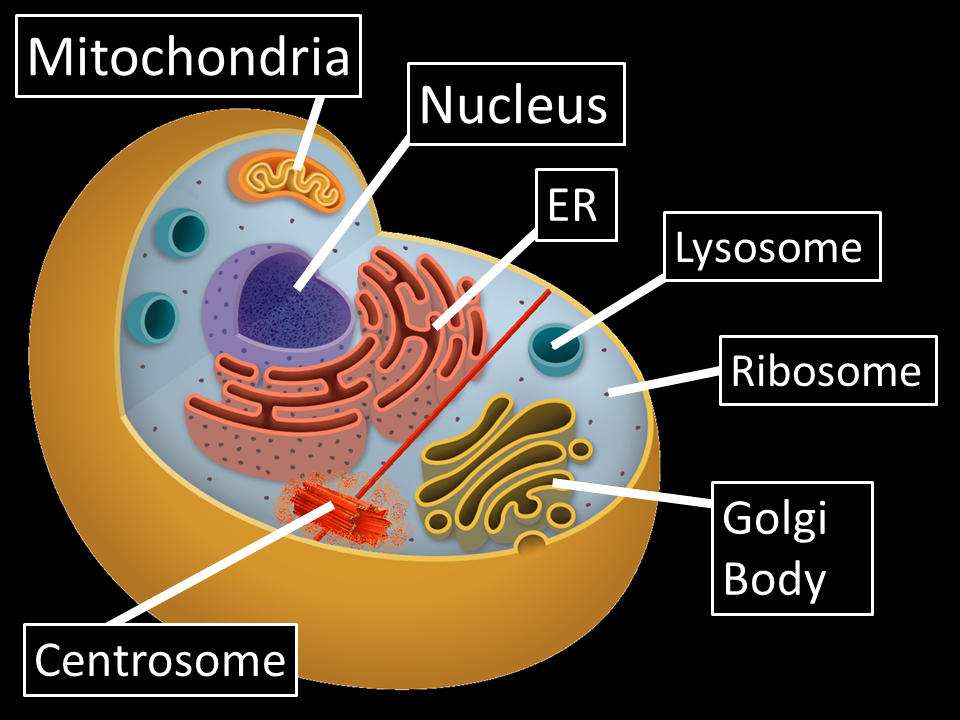
Where Proteins Are Made
Many people wonder about the human body and how it works. Here is some information about how protein is made in the cell and a way to understand it in simple terms.
Proteins are a series of complex molecules. They are needed by the body in a number of ways. The proteins are needed by the cells to help give them a structure, help with the functions of the cell, and assist with the building of tissues in the body as well as the tissues in the organs. Proteins are made up of units called amino acids. These acids form in a long chain . With this chain, they are able to be attached together. Each contains has 20 amino acids. They can be arranged in a number of different ways and combinations to form the proteins. The arrangement of the amino acids will help determine the structure that the protein. This will also help determine the function that the protein is going to be performed in the body. Different proteins will perform different functions.
Proteins are made in the ribosome of the cell. The ribosome is a string of amino acids that are formed together. They are able to use these amino acids to make the proteins. Amino acids work with the mRNA and the tRNA to help make the proteins so the body can use them.
An important job of the proteins is to transport small molecules and other information including atoms in the cells. This will help send messages around the body.
Role Of Protein Synthesis In Disease
Many diseases are caused by mutations in genes, due to the direct connection between the DNA nucleotide sequence and the amino acid sequence of the encoded protein. Changes to the primary structure of the protein can result in the protein mis-folding or malfunctioning. Mutations within a single gene have been identified as a cause of multiple diseases, including sickle cell disease, known as single gene disorders.
Read Also: Vital Proteins Collagen Creamer Review
Signal Sequences Were First Discovered In Proteins Imported Into The Rough Er
Signal sequences were first discovered in the early 1970s in secreted proteins that are translocated across the as a first step toward their eventual discharge from the cell. In the key experiment, the encoding a secreted protein was translated by ribosomes . When microsomes were omitted from this , the protein synthesized was slightly larger than the normal secreted protein, the extra length being the N-terminal leader peptide. In the presence of microsomes derived from the rough ER, however, a protein of the correct size was produced. These results were explained by the signal hypothesis, which postulated that the leader serves as an that directs the secreted protein to the ER membrane and is then cleaved off by a in the ER membrane before the chain has been completed .
The signal hypothesis. A simplified view of protein translocation across the ER membrane, as originally proposed. When the ER signal sequence emerges from the ribosome, it directs the ribosome to a translocator on the ER membrane that forms a pore in
Cell-free systems in which proteins are imported into microsomes have provided powerful assay procedures for identifying, purifying, and studying the various components of the molecular machinery responsible for the import process.
A Possible Role For The Nuclear Turnover In Proof
Finally, we examined what use the cell might make of this apparently-wasteful turnover. As a nuclear ribosome might proof-read nascent RNA for PTCs , turnover could reflect the destruction of peptides produced as a by-product. A test Cd2 gene ± PTC was expressed using a multi-copy system on transfection, the vector encoding Cd2 replicates to generate several thousands of mini-chromosomes that are co-transcribed by cellular polymerases in discrete foci . Cd2 expression is driven by a promoter normally switched off by a modified Tet repressor , but it can be switched on by doxycycline then, levels of nascent Cd2 RNA are monitored by fluorescence in situ hybridization using probes targeting intronic regions of the RNA. As expected, the PTC¯ vector expresses high levels of these intronic regions in many nucleoplasmic foci . The PTC+ vector yields less nuclear signal presumably, because the RNA has been destroyed by NMD. Significantly, cycloheximide increases signal above that seen in the control consistent with it preventing ribosomes from detecting nascent PTC+ transcripts . This places active ribosomes close to pre-mRNA which is found only in nuclei. ].
Cd2
Also Check: Examples Of High Protein Breakfast
Phospholipid Exchange Proteins Help To Transport Phospholipids From The Er To Mitochondria And Peroxisomes
As discussed in Chapter 13, the and the membranes of the Golgi apparatus, lysosomes, and endosomes all form part of a membrane system that communicates with the by means of transport vesicles that transfer both proteins and lipids. Mitochondria, plastids, and possibly peroxisomes, however, do not belong to this system, and they therefore require different mechanisms for the import of proteins and lipids for growth. We have already seen that most of the proteins in these organelles are imported from the . Although mitochondria modify some of the lipids they import, they do not synthesize lipids from scratch instead, their lipids have to be imported from the ER, either directly, or indirectly by way of other cell membranes. In either case, special mechanisms are required for the transfer.
Water-soluble carrier proteins called transfer individual phospholipid molecules between membranes. Each exchange recognizes only specific types of phospholipids. It functions by extracting a of the appropriate phospholipid from a and diffusing away with the buried within its lipid-. When it encounters another membrane, the exchange protein tends to discharge the bound phospholipid molecule into the new . It has been proposed that phosphatidylserine is imported into mitochondria in this way, where it is then decarboxylated to yield phosphatidylethanolamine. Phosphatidylcholine, by contrast, is imported intact.
Trna Molecules Match Amino Acids To Codons In Mrna
The codons in an do not directly recognize the amino acids they specify: the group of three nucleotides does not, for example, bind directly to the . Rather, the translation of mRNA into depends on adaptor molecules that can recognize and bind both to the and, at another site on their surface, to the amino acid. These adaptors consist of a set of small molecules known as transfer RNAs , each about 80 nucleotides in length.
We saw earlier in this chapter that molecules can fold up into precisely defined three-dimensional structures, and the molecules provide a striking example. Four short segments of the folded tRNA are double-helical, producing a that looks like a cloverleaf when drawn schematically . For example, a 5-GCUC-3 sequence in one part of a polynucleotide chain can form a relatively strong association with a 5-GAGC-3 sequence in another region of the same molecule. The cloverleaf undergoes further folding to form a compact L-shaped structure that is held together by additional hydrogen bonds between different regions of the molecule .
A tRNA molecule. In this series of diagrams, the same tRNA moleculein this case a tRNA specific for the amino acid phenylalanine is depicted in various ways. The cloverleaf structure, a convention used to show the complementary
Recommended Reading: Muscle Milk Gainer Protein Powder
What Do Proteins Do For The Body
Our bodies are made up of thousands of different proteins, each with a specific function. They make up the structural components of our cells and tissues as well as many enzymes, hormones and the active proteins secreted from immune cells .
These body proteins are continually being repaired and replaced throughout our lives. This process requires a continuous supply of amino acids. Although some amino acids can be recycled from the breakdown of old body proteins, this process is imperfect. This means we must eat dietary protein to keep up with our bodys amino acid demand.
As protein is essential for cell and tissue growth, adequate intake of protein is particularly important during periods of rapid growth or increased demand, such as childhood, adolescence, pregnancy, and breastfeeding.1
Figure 1. Functions of proteins in the body.
Elongation Factors Drive Translation Forward
The cycle of elongation shown in outline in has an additional feature that makes translation especially efficient and accurate. Two elongation factors enter and leave the during each cycle, each hydrolyzing GTP to GDP and undergoing conformational changes in the process. Under some conditions, ribosomes can be made to perform synthesis without the aid of the elongation factors and GTP hydrolysis, but this synthesis is very slow, inefficient, and inaccurate. The process is speeded up enormously by coupling conformational changes in the elongation factors to transitions between different conformational states of the ribosome. Although these conformational changes in the ribosome are not yet understood in detail, some may involve rearrangements similar to those occurring in the RNAs of the . The cycles of association, GTP hydrolysis, and dissociation ensures that the conformational changes occur in the forward direction and translation thereby proceeds efficiently .
Detailed view of the translation cycle. The outline of translation presented in Figure 6-65 has been supplemented with additional features, including the participation of elongation factors and a mechanism by which translational accuracy is improved.
Don’t Miss: Are Pure Protein Bars Healthy
Formation Of Covalent Bonds
Many proteins produced within the cell are secreted outside the cell to function as extracellular proteins. Extracellular proteins are exposed to a wide variety of conditions. In order to stabilize the 3D protein structure, covalent bonds are formed either within the protein or between the different polypeptide chains in the quaternary structure. The most prevalent type is a disulfide bond . A disulfide bond is formed between two cysteine amino acids using their side chain chemical groups containing a Sulphur atom, these chemical groups are known as thiol functional groups. Disulfide bonds act to stabilize the pre-existing structure of the protein. Disulfide bonds are formed in an oxidation reaction between two thiol groups and therefore, need an oxidizing environment to react. As a result, disulfide bonds are typically formed in the oxidizing environment of the endoplasmic reticulum catalyzed by enzymes called protein disulfide isomerases. Disulfide bonds are rarely formed in the cytoplasm as it is a reducing environment.
Oligosaccharides Are Used As Tags To Mark The State Of Protein Folding
It has long been debated why is such a common modification of proteins that enter the . One particularly puzzling observation has been that some proteins require N-linked glycosylation for proper folding in the ER, yet the precise location of the oligosaccharides attached to the ‘s surface does not seem to matter. A clue to the role of glycosylation in protein folding came from studies of two ER chaperone proteins that are called calnexin and calreticulin because they require Ca2+ for their activities. These chaperones are lectins that bind to oligosaccharides on incompletely folded proteins and retain them in the ER. Like other chaperones, they prevent incompletely folded proteins from undergoing irreversible aggregation. Both calnexin and calreticulin also promote the association of incompletely folded protein with another ER chaperone, which binds to cysteines that have not yet formed disulfide bonds.
Calnexin and calreticulin recognize N-linked oligosaccharides that contain a single terminal , and therefore bind proteins only after two of the three glucoses that are initially attached have been removed by glucosidases. When the third glucose is removed, the dissociates from its chaperone and can leave the ER.
The role of N-linked glycosylation in ER protein folding. The ER-membrane-bound chaperone protein calnexin binds to incompletely folded proteins containing one terminal glucose on N-linked oligosaccharides, trapping the protein in the ER. Removal of the
Recommended Reading: Cookies And Cream Protein Bars