Homogeneous Solution For Gpcr Assays
Cell-based assays have become an indispensable method for screening and compound profiling in the early drug discovery process. To date, such assays have proven to be some of the most reliable and reproducible methods in receptor characterization studies, primary screening campaigns and compound profiling programs. For Gq-coupled GPCR targets specifically, homogeneous fluorescent calcium flux assays with masking technology are the methodology of choice.
Combining a novel fluorophore and proven masking technology, the FLIPR Calcium 5 Assay Kit delivers reliable pharmacology, a larger signal window, and improved assay performance. With the FLIPR Calcium 5 Assay Kit and the FLIPR System, consistent screening of a variety of receptors and targets, especially those with small calcium signal responses, can be obtained in an easy-to-use, homogeneous format.
Read Application Note:
The Many Roles Of G Proteins
G proteins, also known as heterotrimeric proteins, have a single,, and subunit. Their primary function is to transduce signals from the vast majority of receptors to effector systems, and they are only loosely linked to neurotransmitter receptors. membrane attachment and function are regulated by G-proteins. G proteins heterotrimeric nature allows for dynamic attachment and function regulation in the membrane.
G Knockout Mice And G Protein Mutants And Chimeras
Other molecular approaches to investigate the varied roles of G proteins include generating G protein knockout mice or overexpressing a variety of G mutants and chimeras in cell lines and examining their effects in different cellular contexts.
An alternative approach to exploring G protein signals has been to co-express G protein mutants or chimeras in different cell types. Gq chimeric mutants containing Gi or Go tails co-expressed in COS-7 cells with opioid receptors and stimulated with opioid agonist are insensitive to pertussis toxin catalyzed ADP-ribosylation demonstrating an inability of Gi or Go tails to serve as pertussis toxin substrates . Gi-coupled opioid receptors increase Gq signals as demonstrated by the co-expression of constitutively active G mutants in COS-7 cells and requires activated phospholipase beta and G dimers .
You May Like: Protein In Whole Wheat Bread
Two Classes Of Gpcrs: Positive And Negative Regulators
G protein-coupled receptors are involved in a variety of physiological processes, including heart rate regulation and blood pressure regulation. They play an important role in the pathophysiology of many diseases, in addition to obesity, type 2 diabetes, and depression. As the demand for therapeutic drugs targeting G proteins grows, it is becoming more critical than ever to comprehend the structure and function of these critical proteins. Recent research has revealed that GPCRs can be classified into two functional categories: positive and negative regulators. When a positive regulator, such as the 1-adrenergic receptor , binds to agonists and activate G proteins, downstream effector proteins are activated. Serotonin 5-HT1A antagonists, for example, bind and inhibit the activation of G proteins, resulting in the inhibition of effector proteins. G-protein coupled receptors are being studied in order to gain a better understanding of the mechanisms by which many diseases are caused, and their study is likely to be an important part of the development of new therapeutic interventions.
Signaling Without G Proteins

GPCR activation can evoke signal pathways that do not require coupling to G proteins. Much as activated receptor conformations are recognized by their complementary G proteins , so too they can be recognized by other signaling proteins including the GRKs, arrestins, JAK, Src family kinases, and PDZ-domain containing proteins . Such coupling can evoke clathrin-mediated internalization, activation of MAP kinase cascade signaling, or stimulation of Na+/H+ exchange.
Don’t Miss: Does Protein Help Lose Weight
How G Proteins Work
- In the inactive state, Gα has GDP in its binding site.
- When a hormone or other ligand binds to the associated GPCR, an allosteric change takes place in the receptor .
- This triggers an allosteric change in Gα causing
- GDP to leave and be replaced by GTP.
- GTP activates Gα causing it to dissociate from GβGγ .
- Activated Gα in turn activates an effector molecule.
In a common example , the effector molecule is adenylyl cyclase – an enzyme in the inner face of the plasma membrane which catalyzes the conversion of ATP into the “second messenger” cyclic AMP .
Activated Gα is a GTPase so it quickly converts its GTP to GDP. This conversion, coupled with the return of the Gβ and Gγ subunits, restores the G protein to its inactive state.
G Protein Coupled Receptor In Detail
GPCR full form G-protein-coupled receptors are the biggest and most diversified collection of membrane receptors in eukaryotes. These cell surface receptors act like an inbox for communications in the form of light energy, peptides, lipids, carbohydrate, and proteins. Cells receive these messages to alert them of the presence or absence of life-sustaining light or nutrients in their surroundings, or to relay information from other cells. They are also known as seven–transmembrane domain receptors, 7TM receptors, heptahelical receptors, serpentine receptors, and G protein-linked receptors , which are cell surface receptors that detect chemicals outside the cell and activate physiological responses. They are known as seven transmembrane receptors because they bind to G proteins and cross through the cell membrane seven times.
GPCRs are involved in a wide range of processes in the human body, and a better understanding of these receptors has had a significant impact on modern medicine. In fact, experts believe that GPCRs are involved in the action of one-third to half of all marketed medications.
Read Also: Core Power Protein Shake 42g Nutrition Facts
Specificity To G Proteins
In general, GAPs tend to be pretty specific for their target G proteins. The exact mechanism of target specificity is not fully known, but it is likely that this specificity comes from a variety of factors. At the most basic level, GAP-to-G protein specificity may come simply from the timing and location of protein expression. RGS9-1, for example, is specifically expressed in the rod and cone photoreceptors in the eye retina, and is the only one to interact with G proteins involved in phototransduction in this area. A certain GAP and a certain G protein happen to be expressed in the same time and place, and that is how the cell ensures specificity. Meanwhile, scaffold proteins can also sequester the proper GAP to its G protein and enhance the proper binding interactions. These binding interactions may be specific for a particular GAP and G protein. Also, GAPs may have particular amino acid domains that recognize only a particular G protein. Binding to other G proteins may not have the same favorable interactions, and they therefore do not interact. GAPs can, therefore, regulate specific G proteins.
U73122 And Its Inactive Analog U
To maximize our knowledge of Gq, it is possible to examine the downstream role of Gq in GPCR by assessing the inhibitors of PLC, U73122, and its inactive analog U73343. U73122 and its analog U73343 were used to show the effect of human platelet calcium signaling and protein tyrosine phosphorylation in the presence of thrombin, collagen, and thapsigargin . U73122 showed complete inhibition of calcium signaling in the presence of this agonist, which was generated via the activation of PLC specifically the and isoforms . U73343 did not show any calcium inhibitory effect via the activation of PLC but rather showed the calcium inhibitory effect via the upstream activation of cPLA2 in the presence of thapsigargin and collagen . This provides a clear indication that U73343 has minimal activity as a PLC inhibitor.
The study also investigated the role of Gq in the transactivation of PTK receptors to show that platelets, stimulated by thrombin increased protein tyrosine phosphorylation. In the presence of U73122, the phosphorylation of tyrosine kinase was abolished . As mentioned earlier, Gq are prominent in signaling in VSMCs. From the recent study, it is possible that we can replicate this investigation in other cell type to further study the PTK and perhaps the serine/threonine kinase transactivation pathway in the presence of U73122.
Don’t Miss: What Has Protein Besides Meat
Origin And Diversification Of The Superfamily
Signal transduction mediated by the superfamily of GPCRs dates back to the origin of multicellularity. Mammalian-like GPCRs are found in fungi, and have been classified according to the GRAFS classification system based on GPCR fingerprints. Identification of the superfamily members across the eukaryotic domain, and comparison of the family-specific motifs, have shown that the superfamily of GPCRs have a common origin. Characteristic motifs indicate that three of the five GRAFS families, Rhodopsin, Adhesion, and Frizzled, evolved from the Dictyostelium discoideum cAMP receptors before the split of opisthokonts. Later, the Secretin family evolved from the Adhesion GPCR receptor family before the split of nematodes. Insect GPCRs appear to be in their own group and Taste2 is identified as descending from Rhodopsin. Note that the Secretin/Adhesion split is based on presumed function rather than signature, as the classical Class B is used to identify both in the studies.
What Is The G Protein Receptor Pathway
G Protein Concentrating receptors are receptors that sense and transphosphorylate many extracellular signals and send them to heterotrimeric G proteins, which are then converted to appropriate downstream effectors and thus play a variety of roles in various signaling pathways.
Activating heptahelical receptors at the cell surface is accomplished by activating hepta proteins that are found in tandem with intracellular signaling cascades. G protein receptors generate receptor-gated GTP for GDP exchange between G protein alpha subunits and are molecular switches. This chapter describes what we know about receptor and G protein structure, as well as what we know about the receptor-G protein complex and how we can create improved models based on the current data. The Thromb J 2019 study shows that B-cell clonogenic activity of HIV-1 p17 variants is driven by activation of the EGF membrane by the PAR1-mediated pathway. The evolution of heterotrimeric G protein subunits, which was explained by Giagulli C, Caccuri F, Zorzan S, Bugatti A, Zani A, Filippini F, Manocha E, DUrsi P, Orro A, Dolcetti R
Read Also: Halo Top Ice Cream Protein
What Do Gpcrs Do
As their name implies, GPCRs interact with G proteins in the plasma membrane. When an external signaling molecule binds to a GPCR, it causes a conformational change in the GPCR. This change then triggers the interaction between the GPCR and a nearby G protein.
G proteins are specialized proteins with the ability to bind the nucleotides guanosine triphosphate and guanosine diphosphate . Some G proteins, such as the signaling protein Ras, are small proteins with a single subunit. However, the G proteins that associate with GPCRs are heterotrimeric, meaning they have three different subunits: an alpha subunit, a beta subunit, and a gamma subunit. Two of these subunits alpha and gamma are attached to the plasma membrane by lipid anchors .
et al.Nature420,
Ii G Protein Structure
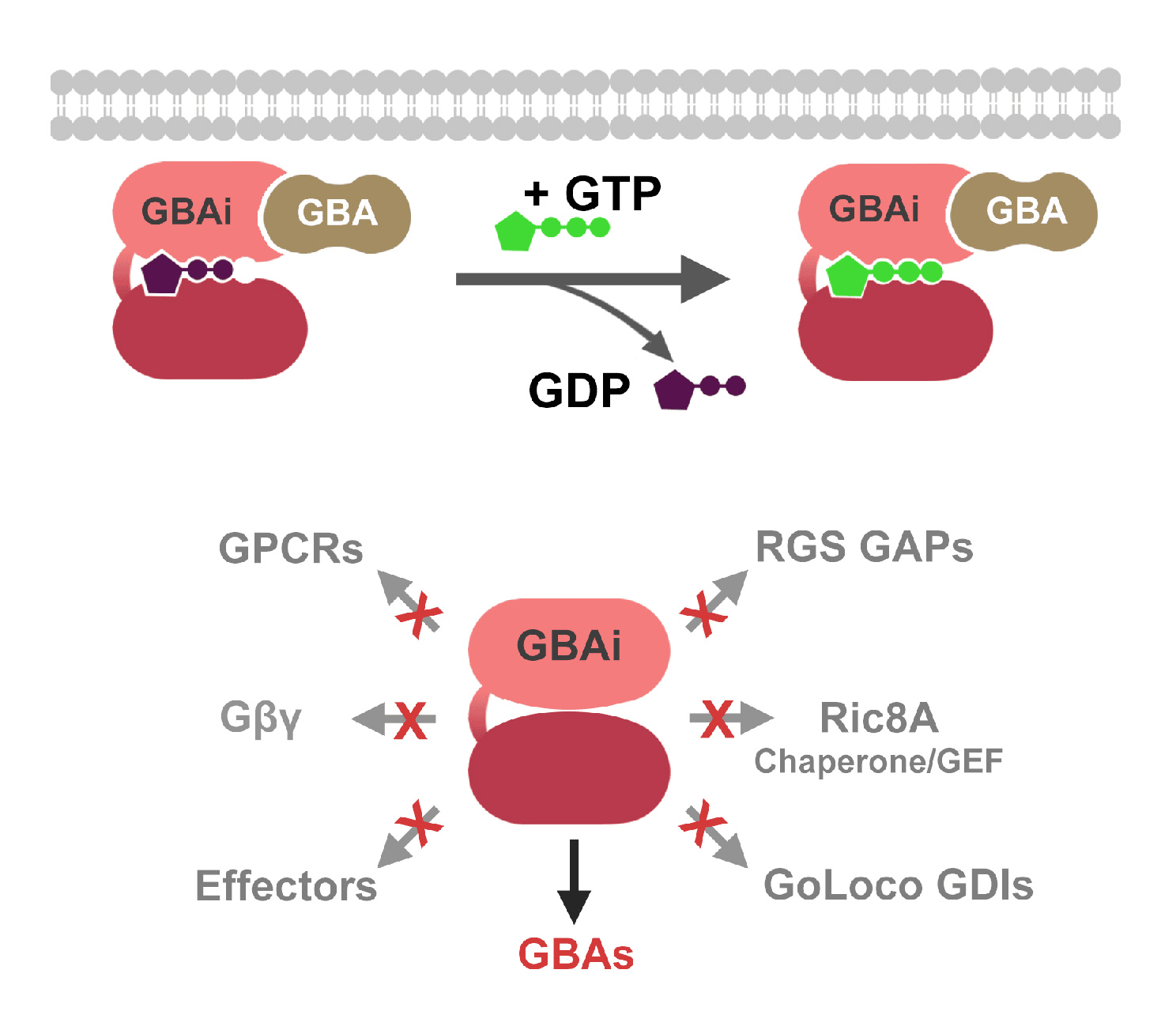
The solution of crystal structures for inactive , active , and transition state Gt or Gi , as well as structures for the inactive heterotrimeric complexes , has provided the framework for understanding the biomechanics of G proteins as molecular switches. For a detailed discussion of the specific intramolecular contacts within heterotrimeric G proteins, the reader is referred to reviews in Refs. and . Herein, we will briefly discuss key structural and functional features common to all heterotrimeric G proteins to understand the framework and interpretation of recent work in this field.
The G-subunit of heterotrimeric G proteins has a -propeller structure containing seven WD-40 repeats . The G-subunit interacts with the G-subunit through an N-terminal coiled coil and makes extensive contacts along the base of the G-subunit . The G-dimer binds to a hydrophobic pocket present in G-GDP. GTP binding to G removes the hydrophobic pocket and reduces the affinity of G for G .
You May Like: How To Get Protein Vegan
Vserotonylation Of Guanine Nucleotide Binding Proteins
G proteins are a major target of serotonylation and other forms of monoaminylation. G proteins include the superfamily of Ras small G proteins, heterotrimeric G proteins, proteins involved in protein synthesis, as well as some less abundant families of G proteins . The Ras superfamily of small G proteins is divided into five main subfamilies, specifically Rho, Rab, Arf, Ran, and Ras based on structure and function. To date, monoaminylation has been reported for members of the Rho and Rab families of small G proteins and G proteins of the heterotrimeric G protein family .
Shlomo Melmed MB ChB, MACP, inWilliams Textbook of Endocrinology, 2020
Grk Activity And Regulation
GRKs reside normally in an inactive state, but their kinase activity is stimulated by binding to a ligand-activated GPCR . Because there are only seven GRKs but over 800 human GPCRs, GRKs appear to have limited phosphorylation site selectivity and are regulated primarily by the GPCR active state.
G protein-coupled receptor kinases phosphorylate activated G protein-coupled receptors, which promotes the binding of an arrestin protein to the receptor. Phosphorylated serine and threonine residues in GPCRs act as binding sites for and activators of arrestin proteins. Arrestin binding to phosphorylated, active receptor prevents receptor stimulation ofheterotrimeric G protein transducer proteins, blocking their cellular signaling and resulting in receptor desensitization. Arrestin binding also directs receptors to specific cellular internalization pathways, removing the receptors from the cell surface and also preventing additional activation. Arrestin binding to phosphorylated, active receptor also enables receptor signaling through arrestin partner proteins. Thus the GRK/arrestin system serves as a complex signaling switch for G protein-coupled receptors.
Read Also: Which Meat Has The Highest Protein
G Protein Signal Transduction
Cell communication through signal transduction is the primary mode of communication between the cells extracellular machinery and its internal organs. G-protein signaling, in particular, plays an important role in the response to hormonal signals as well as environmental stimuli such as light and odor.
G proteins are switches that regulate information processing in information networks by binding to heterotrimeric guanine nucleotides. G proteins play an important role in the metabolic, humoral, neural, and developmental functions of all eukaryotic cells. A number of receptors and effectors have been described in addition to a number of receptors and effectors. PRP4 Gene of S CEREVISIAE, which is a modified version of P21RAS, is related to BETA-subunits of G-proteins, CELL 58: 811 . DERETIC, D., AND OTHER SOCIETY ANALYSIS OF ANTIGENIC DETERMINEANTS RECOGNIZED BY MONOCLONAL-ANNOUNCING BAPTURE-BINDING PROTEIN, TRANSDUCIN, AND MONECOLANICAL DETERMINE An alpha-subunit is an alpha-duplex that links a physiological site to an alpha-duplex site for photosynthesis. The use of GS-ALPHA to alter the active and passive phases of batetine at ARG187 results in a loss of GTPASE activity, as stated by researchers in the Journal of Biological Chemistry. G PROTEAUN DIVERSITY IS EXPIRED BY VALIDATION WITH GAMMA-WHOLES AS PARTNERS.
G Protein Receptor Physiological Roles
GPCRs have a role in a number of physiological processes. The following are some examples of their physiological roles:
The Visual Sense: Opsins convert electromagnetic radiation into cellular messages through a photoisomerization event. Rhodopsin, for example, accomplishes this by converting 11-cis-retinal to all-trans-retinal. Opsins convert electromagnetic radiation into cellular messages through a photoisomerization event. Rhodopsin, for example, accomplishes this by converting 11-cis-retinal to all-trans-retinal.
The Gustatory Sense : Gustducin is released by GPCRs in taste cells in reaction to bitter, umami, and sweet-tasting stimuli.
The Sense of Smell: The olfactory epithelium has receptors that bind odorants and pheromones .
Behavioral and Mood Regulation: Serotonin, dopamine, histamine, GABA, and glutamate are among the neurotransmitters bound by receptors in the mammalian brain.
Regulation of Immune System Activity and Inflammation: Histamine receptors bind inflammatory mediators and engage target cell types in the inflammatory response chemokine receptors bind ligands that promote intercellular communication between immune system cells. GPCRs play a role in immunological modulation as well, controlling interleukin induction and inhibiting TLR-induced immune responses in T cells, for example.
Don’t Miss: Food With Fat And Protein No Carbs
How Does G Protein Function As A Receptor
The majority of external stimuli are mediate by G proteins and receptors that bind together to form proteins, according to cellular biologists. When activated by a ligand, the receptor binds to a partner heterotrimeric G protein and promotes GTP exchange for GDP, resulting in the dissociation of the G protein into subunits and downstream signals.
The Peptide Antagonist Gp
In 2004, Tanski et al. discovered a competitive Gq inhibitor, G Protein antagonist-2A, also known as GP-2A. GP-2A is a peptide that selectively inhibits the action of Gq by M1 muscarinic cholinergic receptors. The signaling pathway of Gq and its role in cell proliferation with rat pulmonary artery smooth muscle cells were studied. Angiotensin II-mediated proliferation, PLC activation, and Erk1/2 phosphorylation were inhibited by more than 50% in the presence of GP-2A . The EGFR can be activated by EGF to generate an intracellular signaling pathway leading to the phosphorylation of several downstream effector proteins such as Erk1/2 . Tanski and colleagues have evaluated angiotensin II to effectively reduce Erk1/2 activation mediated by PLC via Gq in the presence of GP-2A by showing its association with the phosphorylation of Erk1/2 in rat pulmonary artery smooth muscle cells . This study provides a strong foundation for our laboratory research as we can further investigate the possibility of this downstream signaling pathway to see whether or not GP-2A can act on other GPCR agonists such as thrombin to effectively respond similarly via Gq in other smooth muscle cell types such as human VSMCs.
Don’t Miss: High Level Of Protein In Urine
Iii Molecular Basis For G Protein Activation
The rate-limiting step in G protein activation is the release of GDP from the nucleotide-binding pocket. GDP is spontaneously released from the heterotrimeric G protein at a rate that varies depending on the G-subunit. For example, the Go GDP release rate is 0.19 min1 whereas the Gi2 release rate is 0.072 min1 . However, the inactive state of the G-subunits is controlled by G binding. Higashijima et al. showed that in the absence of Mg2+, G increases the affinity of Go for GDP about 300-fold. GDP release is greatly facilitated by receptor activation of the G protein . Mutations of residues in the critical TCAT guanine nucleotide-binding motif present in the 6-5 loop of the GTPase domain enhance receptor-independent spontaneous GDP release. Iiri et al. identified such an activating mutation in Gs in male patients with pseudohypoparathyroidism and gonadotropin-independent precocious puberty. Enhanced GDP release was also observed when similar mutations were generated in Gi and Go suggesting that this region serves as a common mediator of GDP release. Posner et al. also demonstrated that GDP release can occur without inducing a large conformational change in G.